
Interest in the microbiome has expanded significantly over the last decade. However, it is increasingly evident that merely modulating the microbiota may not be sufficient. A more pertinent question is how particular ingredients affect microbial metabolism and whether those modifications produce biologically relevant signals.
A recent paper published in Food Bioscience investigated this interaction using Csat®, a standardized formulation derived from carob, within a controlled in vitro model based on human fecal microbiota. The aim was not to evaluate appetite or body weight outcomes, but rather to gain deeper insight into how Csat® modulates microbial activity and short-chain fatty acid (SCFA) synthesis.
A Closer Examination at SCFAs
Acetate, propionate, and butyrate are key metabolites generated when intestinal bacteria ferment non-digestible substrates. Scientific literature has linked these molecules to several physiological mechanisms, including activation of FFAR2 and FFAR3 receptors and regulation of hormones such as GLP-1 and PYY. The connection between SCFAs and host metabolism is multifaceted and highly context-dependent. Therefore, mechanistic models serve as valuable tools to clarify upstream microbial and metabolic processes prior to clinical investigation.
Study Design and Evaluation
In this work, researchers incubated human gut microbiota under anaerobic conditions and compared Csat® with a control medium as well as two individual carob fractions (a galactomannan-rich seed fraction and a pulp extract).
They evaluated:
-
Variations in microbial composition (full-length 16S rRNA sequencing)
-
SCFA levels over time (quantified by GC-FID)
-
Predicted functional capacity using bioinformatic platforms (PICRUSt2, COG, and CAZy databases)
What Changed with Csat®
After 24 hours, Csat® showed higher SCFA production (Figure 1) compared to control:
-
Acetate: 71.5 mM vs 51.8 mM
-
Propionate: 52.8 mM vs 33.7 mM
-
Butyrate: 48.6 mM vs 34.3 mM
Notably, although the galactomannan-rich fraction (GM) and the pulp extract (CPE) each triggered fermentation activity, Csat® consistently produced the highest and most sustained concentrations of the three primary SCFAs, particularly at 48 hours.
GM displayed strong early stimulation of carbohydrate-degrading potential, while CPE induced a broader yet more short-lived microbial response. By contrast, Csat® integrated these complementary actions, maintaining targeted enrichment of Bacteroides species and supporting butyrate-associated genera such as Anaerostipes and Blautia. At the same time, lower isovalerate levels under Csat® indicated a metabolic shift away from proteolytic fermentation toward sustained carbohydrate-driven metabolism.
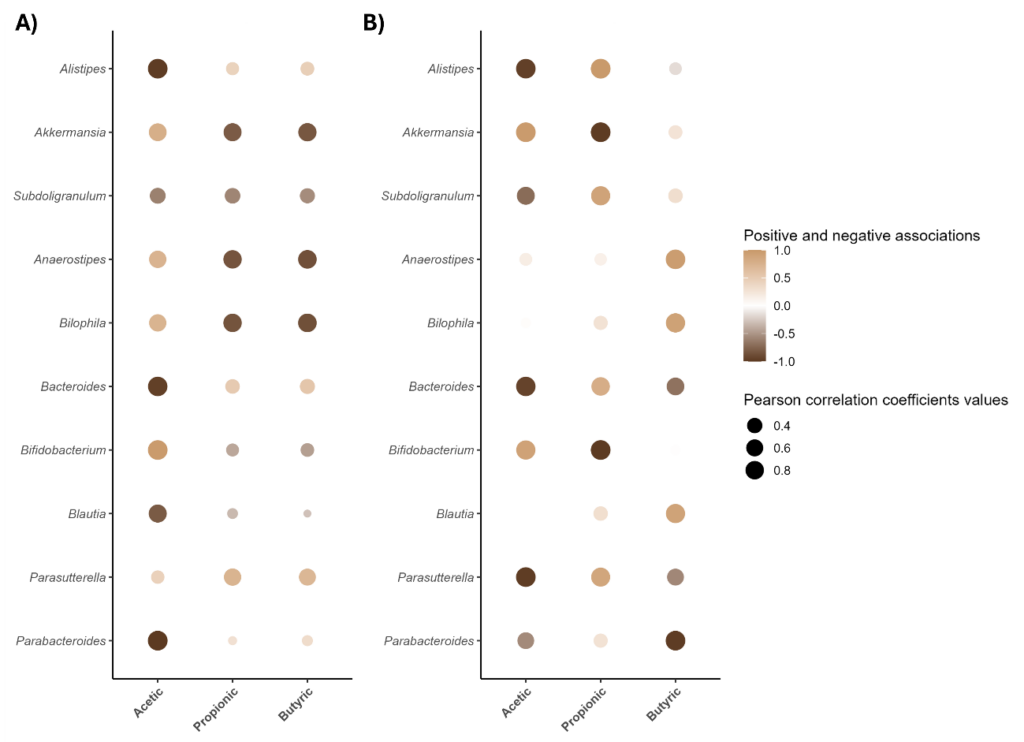
Figure 1. Short-chain fatty acid (SCFA) concentrations (mM) in control and treatment groups (Csat®, GM, and CPE) after 24 h and 48 h of fermentation. Data are shown as mean ± SD (n = 3). Statistical differences were assessed by one-way ANOVA followed by Tukey’s post hoc test (p < 0.05). Different letters above bars indicate significant differences among groups within each time point.
Correlation analyses (figure 2) further suggested coordinated interactions between primary degraders and secondary fermenters under Csat®, consistent with cross-feeding mechanisms known to sustain SCFA production.
Figure 2. Pearson’s correlation between relative abundance of the 10 most abundant bacterial genera and short-chain fatty acids (SCFAs) during fecal fermentation of Csat®. (A) Correlations at 24 h and (B) at 48 h of fermentation. Circle size represents the absolute value of the correlation coefficient, and color scale indicates the direction and strength of the association (brown = positive, beige = negative). Only significant associations (p < 0.05) are shown.
Functional predictions revealed a temporally aligned pattern. The GM fraction showed early enrichment of glycosidase-related signatures consistent with rapid extracellular breakdown of complex carbohydrates. Conversely, Csat® exhibited a later increase in predicted sugar transport systems, particularly ATP-binding cassette (ABC) transporters, indicating enhanced uptake and intracellular utilization of fermentation products over time.
This integrated functional profile—combining hydrolytic activation with sustained substrate transport—was not observed to the same degree with the isolated fractions. It offers a mechanistic rationale for the higher and more prolonged SCFA production seen under Csat® fermentation.
Although these functional interpretations are based on predictive bioinformatic analyses rather than direct enzymatic measurements, they align with the metabolite outputs and taxonomic shifts identified.
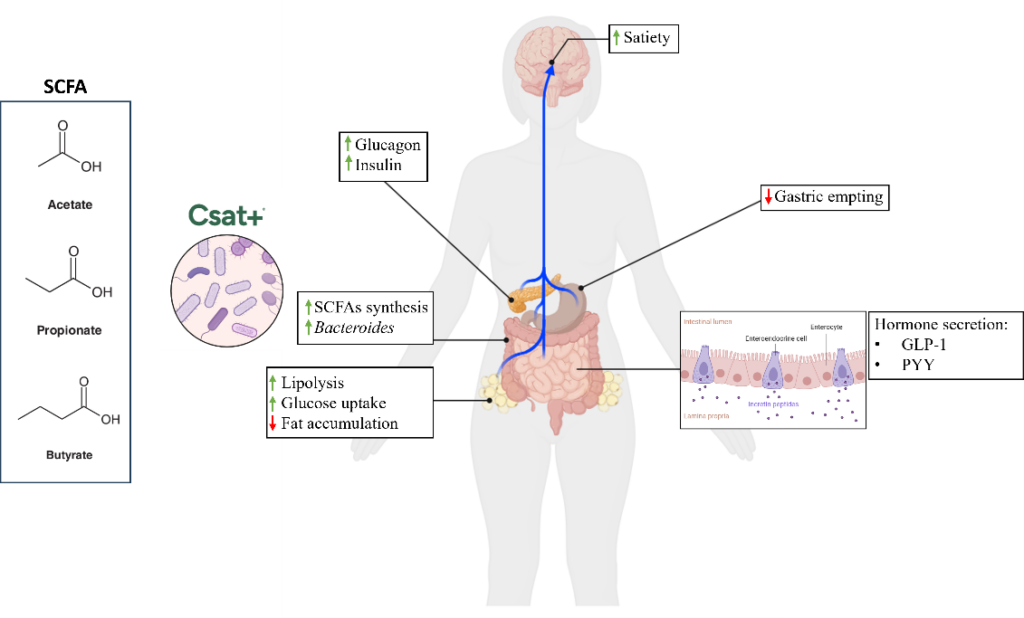
Overall, these results support the biological plausibility of a microbiota-mediated mechanism in which coordinated carbohydrate breakdown, cross-feeding interactions, and efficient substrate utilization converge to enhance SCFA production—an effect mechanistically connected in the broader literature to satiety-related signaling pathways.
Why This Matters
Research in the microbiome field is evolving beyond general claims of microbiota modulation toward a more mechanistic perspective on how defined, standardized ingredients reshape microbial metabolism.
In this framework, Csat® distinguishes itself. Unlike isolated carob fractions, Csat® is a fully characterized and standardized formulation that integrates multiple bioactive components under controlled manufacturing processes. This compositional consistency enables reproducible functional outcomes, as illustrated in the integrated fermentation model.
By combining complementary structural fractions of carob, Csat® facilitates coordinated carbohydrate depolymerization, substrate uptake, and sustained short-chain fatty acid generation. This integrated microbial response is directly linked to biological pathways involved in:
-
Satiety signaling along the gut–brain axis
-
Enteroendocrine hormone regulation (GLP-1, PYY)
-
Energy metabolism and substrate allocation
-
Systemic metabolic homeostasis
Measuring metabolite output, identifying taxonomic changes, and predicting functional capacity creates a mechanistic bridge between ingredient composition and physiological relevance.
In this sense, the study goes beyond simply reporting microbiota modulation; it contributes to a rational, evidence-based framework for designing metabolically active ingredients intended to interact with the gut–brain–metabolic axis.
About the Research
The study was led by Iván Benito Vázquez in collaboration with researchers from the Instituto de Investigación en Ciencias de la Alimentación (CIAL-CSIC) and Pharmactive Biotech Products. This partnership reflects the integration of academic investigation and industrial standardization in the development of scientifically substantiated botanical ingredients.
References
Benito-Vázquez, I., Yépez-Notario, C., Díez-Municio, M., Requena, T., & Moreno, F. J. (2026). Unveiling the microbiome-mediated potential of a carob-based formulation (Csat®): human fecal fermentation, SCFA production, and potential connections with satiety pathways. Food Bioscience, 78, 108472. https://doi.org/10.1016/j.fbio.2026.108472

